Quote:
Originally Posted by atom
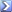
Equation for denitrifying process:
Nitrate to Nitrite to Nitrogen
Redox equation (I prefer)
NO3 + 2 electrons + H+ <=> NO2 + H2O
NO2 + 3 electrons + 4 H+ 1/2 N2 (nitrogen gas) + 2 H2O
or taken together:
NO3 + 5 electrons + 6 H+ <=> 1/2 N2 + 3 H2O
|
Wow... Looks like my chemistry lessons many many many years ago... Anyway, thanks for the info... But I am still debating whether having a denitrator or just changing water is more feasible or make more economic sense..